PHYSICIAN'S GUIDE
TO
PESTICIDE POISONING
Table of Contents | Preface | Section I | Section II | Section III | References
SECTION III
PATIENT MANAGEMENT
The following section will provide a general overview of pesticide poisoning, toxicology, recognition and management by pesticide categories.
Regardless of the poisoning, there are certain precautions that all providers must take with a potential pesticide exposure.
RECOMMENDATIONS FOR PATIENT:
For patients suffering from dermal exposure, wash patient with soap and water immediately.
Transport patient to the nearest doctor, hospital or clinic.
Family member, friend, co-worker, or employer should drive. Patient must not drive. TRANSPORT WITH PESTICIDE LABEL WHENEVER POSSIBLE.
Inform physician of suspected exposure to pesticides.
PHYSICIAN SHOULD REQUEST PESTICIDE LABEL, MATERIAL SAFETY DATA SHEETS (MSDS), and RECORDS. FOR FARM WORKERS, ALSO REQUEST CROP SHEETS.
Physicians and other health professionals have the right to ask employers for any pertinent information. This includes Workplace Chemical Lists, logs, precautions, name of pesticides used, and EPA registration numbers. Above all, call the poison control center for emergency instructions on separate recommendations. Also use pesticide emergency "hotline" from pesticide labels or MSDS.
To avoid exposure or poisoning, workers must strictly follow application and handling instructions. They also must and take many precautions. These precautions are frequently mandated by laws or regulations. When poisoning of farm workers, pesticide applicators, mixers, loaders, and handlers occurs, it is usually because they did not follow the above precautions.
NOTE:
Special attention should be given to children suspected of pesticide poisoning. This can occur through direct contact or ingestion of household chemicals. It also can occur through exposure to residues in the house, on lawns or in gardens. It may occur in family members of farm laborers when workers come in from the field. Or it may occur when home pesticide users come into the house after applying a lawn or garden pesticide. It may occur when contaminated clothing is washed with the family laundry. The NAS (1993) report on pesticides shows that acute and chronic toxicity thresholds can be lower for children than for adults. Antidote recommendations for children and dose levels also require administration accordingly.
For more detailed information on treatments, consult Recognition
and Management of Pesticide Poisoning, 4th Edition by Donald P. Morgan
(1989). Clinical Toxicology of Commercial Products by Gosselin et
al (1984) also may prove useful in diagnosis, treatment, and therapy.
Toxicology
Chemical Effects Exposure
Acetylcholine accumulation is caused by irreversible phosphorylation of the acetylcholinesterase enzyme.
Cholinergic junctions produce muscarinic effect on smooth muscles and gland cells, causing muscle contractions and secretions.
Nicotinic junctions produce excitatory effects on skeletal muscles and autonomic ganglia, but can weaken or paralyze the end plat cells.
Brain - sensory and behavioral disturbance, incoordination and depressed motor function.
(respiratory depression and pulmonary edema are usual causes of death; reported pesticide cases involving children are more likely to be organophosphate poisonings)
inhalation, ingestion, absorption. In some cases organophosphates can be stored in fat cells for prolonged periods. Neurotoxicity in this case can cause weakness, paralysis, paraesthesia of the extremities predominantly of the legs -- persisting for weeks to years.
Symptoms/ Signs
Develop immediately after exposure or within 12 hours (average onset is within 4 hours). Most prominent are:
HEADACHE NAUSEA
DIZZINESS MUSCLE TWITCHING
WEAKNESS HYPERSECRETION
MIOSIS PULMONARY EDEMA
Other symptoms are: anxiety, restlessness, tremor, incoordination, vomiting, abdominal cramps, diarrhea, sweating, salivation, tearing, rhinorrhea, bronchorrhea, blurred or dark vision, chest tightness, wheezing, productive cough, tachycardia, hypertension, sinus arrest, toxic psychosis, confusion, bizarre behavior, unconsciousness, incontinence and convulsions.
A constant exposure at low doses can cause persistent anorexia, weakness,
and malaise. Acute ingestion may cause prolonged paralysis of the head,
neck, limbs and thorax muscles.
Laboratory
DO NOT WAIT FOR LABORATORY CONFIRMATIONS IF THERE ARE STRONG CLINICAL INDICATIONS OF ORGANOPHOSPHATE POISONING.
Test for low cholinesterase levels in plasma or red blood cells. there are various tests available (Michael, Nabb-Whitfield, Ellman-Boehringer). A twenty-five percent or more depression is generally regarded as an exposure/poisoning.
It is important to document baseline or preexposure levels. Many persons have an established level of cholinesterase that will test normal but is actually lower and should be considered a case of poisoning. When in doubt draw two samples 4 weeks apart. A significant change between the two levels is suggestive of poisoning.
Treatment
1. If necessary, clear airway and administer oxygen (lavage may be necessary, as well as cardiac and respiratory mechanical support and monitoring).
2. Atropine sulfate IV (preferred) >12 yrs. 0.4-2.0 mg q/15'(until atropinization: flushing, dry mouth, dilated pupils, and tachycardia/140') <12 yrs. 0.05 mg/kg q/15'
3. Draw heparinized blood sample
4. Pralidoxime (protopam, 2-PAM) may be necessary if severe respiratory or neuromuscular compromise exists. >12 yrs. 1-2 gm/minute IV (No more than 0.2 gm/minute) <12 yrs. 20-50 mg/kg. Repeat in 1-2 hours, then in 10-12 intervals. (Do NOT use for probable carbamate poisoning)
5. 72-hour observation
If ingested, gastric lavage is necessary to prevent central nervous system (CNS) depression.
6. Intubate, aspirate, lavage
7. Remember to protect airway
8. Use large orogastric tube
9. Lavage with activated charcoal in isotonic saline
10. After lavage, instill activated charcoal with a cathartic. >12
yrs. 50-100 gm/300-800 ml water. <12 yrs. 15-30 gm/100-300 ml water
ORGANOPHOSPHATES
The tables below list some common organophosphate insecticides used in crops in various regions of Texas. The tables contain the brand name, common name of active ingredient, crops or sites of application, and the page reference for identification and treatment in the Morgan (1989) manual.
NORTHEAST
| BRAND NAME | CHEMICAL | CROPS | Morgan (1989) |
| DIAZINON | DIAZINON | BLUEBERRIES, MELONS,
COASTAL BERMUDA |
page 1-11. |
| LORSBAN | CHLORPYRIFOS | CORN, SORGHUM | page 1-11. |
| COUNTER | TERBUFOS | CORN | page 1-11. |
| CYGON | DIMETHOATE | CORN, WATERMELONS,
MELONS, WHEAT SORGHUM |
page 1-11. |
| DI-SYSTON 8 | DISULFOTON | CORN | page 1-11. |
| GUTHION | AZINPHOSMETHYL | COTTON, PEACHES,
PECANS |
page 1-11. |
| PENCAP-M | METHYL PARATHION | COTTON, WHEAT,
SOUTHERN PEAS |
page 1-11. |
| PARATHION | ETHYL PARATHION | COTTON, WHEAT,
SORGHUM |
page 1-11. |
| BIDRIN | DICROTOPHOS | COTTON | page 1-11. |
| PHOSDRIN | MEVINPHOS | MELONS | page 1-11. |
| MALATHION | MALATHION | MELONS, COASTAL BERMUDA | page 1-11. |
| DYLOX | TRICHLOROFON | COASTAL BERMUDA | page 1-11. |
SOUTHEAST
| BRAND | CHEMICAL | COMMON CROPS | |
| PENCAP | METHYL PARATHION | WHEAT, SOYBEANS, RICE, COTTON | page 1-11. |
| ORTHENE | ACEPHATE | COTTON | page 1-11. |
| GUTHION | AZINPHOSMETHYL | COTTON, PECANS, PEACHES | page 1-11. |
| PARATHION | ETHYL PARATHION | COTTON, WHEAT, SORGHUM | page 1-11. |
| page 1-11. | |||
| CYGON | DIMETHOATE | WHEAT, WATERMELONS | page 1-11. |
| COUNTER | TERBUFOS | CORN, SORGHUM | page 1-11. |
| ZOLONE | PHOSALONE | PECANS, PEACHES | page 1-11. |
| MALATHION | MALATHION | PECANS, SQUASH | page 1-11. |
| LORSBAN | CHLORPYRIFOS | PECANS, SORGHUM, PEANUTS | page 1-11. |
| DIAZINON | DIAZINON | WATERMELONS, PEANUTS, BLUEBERRIES, COASTAL BERMUDA | page 1-11. |
| TRITHION | CARBOFENTHION | WATERMELONS | page 1-11. |
| DYLOX | TRICHLORFON | COASTAL BERMUDA | page 1-11. |
SOUTH TEXAS and LOWER RIO GRANDE VALLEY
| BRAND | CHEMICAL | COMMON CROPS | Morgan (1989) |
| PHOSDRIN | MEVINPHOS | TURNIPS, SPINACH, CELERY, LETTUCE | page 1-11. |
| DIAZINON | DIAZINON | TURNIPS, ONIONS, COLLARDS, KALE, KOHLRABI, MUSTARD GREENS, SWISS CHARD | page 1-11. |
| METASYSTOX-R | OXYDEMETON-METHYL | SQUASH, CUCUMBERS, MELONS, WATERMELONS | page 1-11. |
| LORSBAN | CHLORPYRIFOS | SORGHUM, CORN, CITRUS | page 1-11. |
| COUNTER | TERBUFOS | SORGHUM, CORN | page 1-11. |
| CYGON | DIMETHOATE | SORGHUM, MELONS, TOMATOES, WATERMELONS, KALE, KOHLRABI, COLLARDS, MUSTARD GREENS, SWISS CHARD | page 1-11. |
| ORTHENE | ACEPHATE | PEANUTS, LETTUCE | page 1-11. |
| DI-SYSTON | PEANUTS, CAULIFLOWER | page 1-11. | |
| page 1-11. | |||
| MONITOR | METHAMIDOPHOS | CAULIFLOWER, MELONS CABBAGE, PEPPERS, WATERMELONS, BROCCOLI | page 1-11. |
| GUTHION | AZINPHOS-METHYL | BROCCOLI, PEPPERS, COTTON, PEACHES, SUGARCANE | page 1-11. |
| PENCAP-M | METHYL PARATHION | COTTON | page 1-11. |
WEST TEXAS and HIGH PLAINS
| BRAND | CHEMICAL | COMMON CROPS | Morgan (1989) |
| PARATHION | ETHYL PARATHION | ALFALFA, COTTON | pp. 1-11. |
| PENCAP-M | METHYL PARATHION | ALFALFA, ONIONS | pp. 1-11. |
| CYGON | DIMETHOATE | ALFALFA, SEED SORGHUM | pp. 1-11. |
| LORSBAN | CHLORPYRIFOS | ALFALFA, SEED SORGHUM | pp. 1-11. |
| MONITOR | METHAMIDOPHOS | MELONS, CABBAGE | pp. 1-11. |
| PHOSDRIN | MEVINPHOS | MELONS, WATERMELONS, PEPPERS | pp. 1-11. |
| pp. 1-11. | |||
| THIMET | PHORATE | SUGAR BEETS | pp. 1-11. |
| ORTHENE | ACEPHATE | PEPPERS | pp. 1-11. |
| BIDRIN | DICROTOPHOS | COTTON | pp. 1-11. |
Toxicology
Acetylcholine accumulation is caused by reversible carboxylation of the acetylcholinesterase enzyme.
Chemical effects
Cholinergic junctions produce muscarinic effects on smooth muscles and gland cells casing muscle contractions and secretions.
Nicotinic effects produce excitatory effects on skeletal muscles and autonomic ganglia that can cause twitching and also, weaken or paralyze end plate cells.
Brain - sensory and behavioral changes, incoordination and depressed motor function.
Note: Unlike organophosphate acetylcholine accumulation, carbamates dissociate more readily, which:
limits duration of poisoning
produces greater span between symptom production and lethal dose
invalidates blood CHE fluids
Exposure
Inhalation, ingestion, dermal. Excreted by the kidneys and liver.
Symptoms/Signs
Early = malaise, muscle weakness, dizziness and sweating.
Other symptoms: headache, salivation, nausea, vomiting, abdominal pain, diarrhea, miosis, incoordination, slurred speech, dyspnea, bronchospasm, chest tightness, pulmonary edema, blurred vision, muscle twitching, spasms, convulsions and cardiac complications.
Laboratory
If blood sample is not drawn within 1-2 hours after exposure; cholinesterase levels will not be depressed.
Urine analysis should be done for N-Methyl Carbamate metabolites.
If clinically strong for acute poisoning, DO NOT WAIT for laboratory confirmation.
Treatment
1. Clear airway, oxygen and gastric lavage may be necessary as well as cardiac and respiratory mechanical support and monitoring.
2. Atropine sulfate IV (preferred)
>12 yrs. 0.4-2.0 mg q/15'
(until atropinization)
<12 yrs. 0.05 mg/kg q/15'
(DO NOT USE PRALIDOXIME)
3. 1. Draw heparinized blood sample
2. Take urine sample for metabolites
4. 72-hour observation
Carbamates (Continued)
If ingested, gastric lavage is necessary to prevent CNS depression.
1. Intubate, aspirate, lavage
2. Remember to protect airway
3. Use large orogastric tube
4. Lavage with activated charcoal in isotonic saline
5. After lavage, instill activated charcoal with a cathartic:
>12 yrs. 50-100 gm/300-800 ml water
<12 yrs. 15-30 gm/100-300 ml water
CARBAMATE INSECTICIDES
The tables below list some common carbamate insecticides used in crops in various regions of Texas. The tables contain the brand name, common name of active ingredient, crops or sites of application, and the page reference for identification and treatment in the Morgan (1989) manual.
NORTHEAST
| CHEMICAL | BRAND | CROPS | Morgan (1989) |
| CARBARYL | SEVIN | BLUEBERRIES, PEACHES
PECANS, SORGHUM, SOUTHERN PEAS, WATERMELONS, WHEAT, COASTAL BERMUDA |
pp. 12-24. |
| CARBOFURAN | FURADAN | CORN, SORGHUM | pp. 12-24. |
| ALDICARB | TEMIK | PEANUTS | pp. 12-24. |
SOUTHEAST
| CHEMICAL | BRAND | CROPS | Morgan (1989) |
| CARBARYL | SEVIN | SQUASH, PECANS, WATERMELONS, CORN PEACHES, RICE, COASTAL BERMUDA, BLUEBERRIES | pp. 12-24. |
| CARBOFURAN | FURADAN | CORN, RICE, SORGHUM | pp. 12-24. |
| MOLINATE | ORDRAM | RICE | pp. 12-24. |
SOUTH VALLEY
| CHEMICAL | BRAND | CROPS | Morgan (1989) |
| METHOMYL | LANNATE | PEPPERS, ONIONS, TOMATOES, LETTUCE, CUCUMBERS, CORN, CAULIFLOWER, CABBAGE, BROCCOLI, SPINACH, CELERY, MUSTARD GREENS, SQUASH KALE, COLLARDS, TURNIPS, SWISS CHARD, KOHLRABI, WATERMELON | pp. 12-24. |
| CARBARYL | SEVIN | PEANUTS, LETTUCE, MUSTARD GREENS, KALE COLLARDS, PEPPERS, PEACHES, SORGHUM, TURNIPS, SWISS CHARD, KOHLRABI | pp. 12-24. |
| CARBOFURAN | FURADAN | SORGHUM, CORN | pp. 12-24. |
| ALDICARB | TEMIK | PEANUTS, CITRUS | pp. 12-24. |
WEST
| CHEMICAL | BRAND | CROPS | Morgan (1989) |
| CARBARYL | SEVIN | ALFALFA, GRAPES | pp. 12-24. |
| METHOMYL | LANNATE | ALFALFA, GRAPES, WATERMELONS, CABBAGE, PEPPERS, POTATOES, SEED SORGHUM | pp. 12-24. |
| CARBOFURAN | FURADAN | ALFALFA, SUGAR BEETS | pp. 12-24. |
ORGANOCHLORINES (Insecticides)
Toxicology
Gastrointestinal absorption or respiratory through aerosols and volatile. Causes interference with fluxes of cations across the nerve cell membranes.
Chemical Effects
Myoclonic Jerking
(Nerve cell membrane interference causes neuronal irritability).
Convulsions
Pulmonary gas exchange interference (metabolic acidosis)
Myocardial
Cardiac arrhythmias.
High concentrations induce hepatic enzyme activity, biotransformation of steroid hormones, lipophilic compounds are likely to be excreted in maternal milk, causes porphyria cutanea and aplastic anemia
Exposure
Absorption by ingestion, dermal, gastrointestinal absorption
Symptoms/Signs
Early onset symptoms of these chemicals are similar to some organophosphates (check symptom section on organophosphates or the Morgan manual for further description)
Sensory disturbance hyperesthesia, paresthesia of face and extremity.
Headache, dizziness, nausea, vomiting, incoordination, tremor, mental
confusion, myoclonic jerking tonic-clonic convulsions.
Organochlorines (Insecticides) Continued
Non-DDT like symptoms cause:
immediate convulsions 48 hours after exposure and continue over days; prolonged chronic exposure = weight loss, tremor, muscle weakness, involuntary eye movement, chest and joint pain, skin rash, slurred speech, mental changes.
Respiratory metabolic acidosis and death.
Laboratory
Blood gas chromatographic exams and urinary metabolites. These are performed through university/poison control centers and private labs
Treatment
Observe for toxicosis; sensory disturbances, incoordination, slurred speech, involuntary motor activity (convulsions).
Take seizure precautions
Oxygen + anticonvulsive
Gastric lavage may be necessary
Monitor and assist pulmonary ventilation and cardiac status.
BIOLOGICAL INSECTICIDES (repellents, pyrethrins, nicotine as acaricides)
Toxicology
Pyrethrins are botanical insecticides with very low mammalian toxicity. They lack environmental persistence and break down rapidly. Commercial pyrethrum is a powder seldom found outside industry. Pyrethrum extract is the usual commercial form. The extract is a preparation of pyrethrins in an organic solvent. The solvent may be methanol, acetone, kerosene, fuel oil, or any other petroleum distillate. Formulations exist as extract alone or in combination with other insecticides. They come in human and veterinary pharmaceuticals, in liquid concentrates, and as aerosols like "bug bombs." Frequently, the carriers and propellants are more toxic than the pyrethrins themselves.
Chemical Effects
Dermal and respiratory (allergens) (anaphylactic in some cases)
Exposure
Skin, pulmonary mucous membranes
Signs/Symptoms
Asthma reactions; allergic rhinitis; contact dermatitis; cholinesterase symptomatology if combined with organophosphates or carbamates.
Laboratory
Will vary between specific biological insecticides.
Biological Insecticides (Continued)
Treatment
Avoid content with the pesticide residues or inhalation of the chemical vapors
Antihistamines
Treat asthmatic symptomatology
Treat chronic dermatitis with steroids
Eye flushing with clean water and saline solution
Treat other toxic effects of organophosphates, carbamates and other insecticides
Treat nicotinic poisoning with decontamination washing
with soap and water, may need to administer pulmonary ventilation
The tables below list some common insecticides used in crops in various regions of Texas. The tables contain the brand name, common name of active ingredient, crops or sites of application, and the page reference for identification and treatment in the Morgan (1989) manual.
| NORTHEAST | BRAND | CHEMICAL | CROPS | Morgan (1989) |
| (SUBCATEGORIES | ||||
| (PYRETHROIDS) | AMMO | CYPERMETHRIN | COTTON, ONIONS | pp. 34-36. |
| ASANA | ESEN-
FENVALERATE |
PECANS, WATERMELONS | pp. 34-36. | |
| AMBUSH, POUNCE | PERMETHRIN | PECANS, VEGETABLES, MUSHROOMS | pp. 34-36. | |
| KARATE | LAMBDA-CYHALOTHRIN | COTTON | pp. 34-36. | |
| BAYTHROID | CYFLUTHRIN | COTTON | pp. 34-36. | |
| SCOUT | COTTON | pp. 34-36. | ||
| (BIOLOGICAL) | DIPEL | Bacillus thuringensis | SWEET POTATOES | pp. 32-33 |
| (ELEMENTAL SULFUR) | DUSTING SULFUR | SULFUR | PEACHES | pp. 48-49 |
| SOUTHEAST | BRAND | CHEMICAL | CROPS | Morgan (1989) |
| (ORGANOCHLORINE | THIODAN | ENDOSULFAN | SQUASH | pp. 17-24 |
| MARLATE | METHOXYCHLOR | pp. 17-24 | ||
| (PYRETHROIDS) | AMMO | CYPERMETHRIN | COTTON | pp. 34-36 |
| PYDRIN | FENVALERATE | COTTON | pp. 34-36 | |
| SCOUT | TRALOMETHRIN | COTTON, SOYBEANS | pp. 34-36 | |
| AMBUSH
POUNCE |
PERMETHRIN | SOYBEANS | pp. 34-36 | |
| (ELEMENTAL SULFUR) | DUSTING SULFUR | SULFUR | PEACHES | pp. 48-49 |
| SOUTH/VALLEY | BRAND | CHEMICAL | CROPS | Morgan (1989) |
| (PYRETHROIDS) | AMBUSH
POUNCE |
PERMETHRIN | SPINACH, TURNIPS, TOMATOES, COLLARDS, MUSTARD GREENS, KALE PEPPERS, LETTUCE, BROCCOLI, CABBAGE, CORN, CELERY, CAULIFLOWER, SWISS CHARD, KOHLRABI | pp. 34-36. |
| PYDRIN | FENVALERATE | SQUASH, CABBAGE | pp. 34-36. | |
| ASANA | ESENFENVALERATE | TOMATOES, CABBAGE, CUCUMBERS, SQUASH | pp. 34-36. | |
| AMMO
CYMBUSH |
CYPERMETHRIN | COTTON, ONIONS | pp. 34-36. | |
| (ORGANOCHLORINE) | KELTHANE | DICOFOL | CITRUS | pp. 17-24 |
| ACARABEN | CHLOROBENZILATE | CITRUS | pp. 17-24 | |
| THIODAN | ENDOSULFAN | CUCUMBERS, TOMATOES | pp. 17-24 | |
| DIPEL | Bacillus thuringensis | COLLARDS, LETTUCE, KALE, CELERY, MUSTARD, GREENS, CABBAGE, BROCCOLI, TURNIPS, SPINACH, SWISS CHARD, KOHLRABI, CAULIFLOWER | pp. 32-33 | |
| (INORGANIC SALT) | KOCIDE | COPPER HYDROXIDE | PEPPERS | pp. 103 |
| TRI-BASIC CuSO4,
C-O-C-S, FIXED COPPER |
CUPRIC OXYCHLORIDE SULFATE | PEPPERS, TOMATOES | pp. 103 |
| WEST | BRAND | CHEMICAL | CROP | Morgan (1989) |
| (PYRETHROIDS) | AMBUSH
POUNCE |
PERMETHRIN | COTTON, MELONS, CABBAGE | pp. 34-36. |
| ASANA | ESENFENVALERATE | COTTON, PEANUTS | pp. 34-36. | |
| PYDRIN | FENVALERATE | COTTON, MELONS, POTATOES | pp. 34-36. | |
| AMMO
CYMBUSH |
CYPERMETHRIN | ONIONS | pp. 34-36. | |
| (INORGANIC) | DUSTING SULFUR | SULFUR | GRAPES | pp. 48-49 |
| (ORGANOCHLORINE) | ENDOCIDE
THIODAN |
ENDOSULFAN | WATERMELONS, PEPPERS, CABBAGE | pp. 17-24 |
| (INORGANIC SALT) | KOCIDE | COPPER HYDROXIDE | PEPPERS | pp. 103 |
Toxicology
Effects vary with rodenticide type and organ affected. These are usually in the form of baits, powders and dusts. Principle types are anticoagulants, inorganics, convulsants, and a variety compounds including single feeding and cumulative poisons.
Anticoagulants (Coumarins and Indandiones depress the hepatic vitamin K dependent synthesis of substances essential to blood clotting: prothrombin (factor II) and factors VII, IX and X. Warfarin (a coumarin) and chlorphacinone (an indandione) can be absorbed across the skin. However, this occurs only under extraordinary conditions. Ordinarily, poisoning by these agents occurs through ingestion.
Inorganic rodenticides include yellow phosphorus, zinc phosphide, and thallium sulfate. Each produce varied effects depending upon which organ is affected.
Zinc phosphide, a common rodenticide used in orchards against voles and meadow mice liberates phosphine gas when acted upon by moisture. The effects may manifest themselves as damage to the liver, lungs, heart, and central nervous system. If the patient survives initial shock from ingestion,
The convulsants are named more from the symptoms they produce than from a common mode of action. Strychnine causes violent epileptiform convulsions by direct excitatory action on cells of the central nervous system. The fluoroacetates are latent inhibitors requiring metabolic conversion in the liver to fluorocitrate which poisons critical enzymes of the Kreb's cycle and impairs cellular respiration.
Substituted ureas: Pyraminil (Vacor®) is a substituted urea compound with high mammalian toxicity. It is a single-dose rodenticide. It is no longer registered in the United States as a rodenticide. However, home stocks of this product still exist and several suicides and accidental poisonings have resulted from this compound. Severe effects have resulted from very small doses. Substituted urea rodenticides are specific poisons for cells of the pancreas. Pyraminil also has direct effect on glucose metabolism. It also produces pronounced neurotoxic effects. The exact mechanism of action of substituted urea rodenticides remains uncertain.
Chemical effects
Inorganic compounds like phosphorus and zinc phosphide are highly reactive and corrosive to tissues. Phosphorus may burn skin. Others may produce system manifestations.
Exposure
Gastrointestinal, skin, mucous membranes
Symptoms/Signs
Anticoagulants:
Coumarins Initial symptoms include increasing pallor, weakness, back pain, abdominal pain, pain in the extremities, and vomiting. Later symptoms include nosebleed, bleeding of mouth and gums, massive bruises, hematoma of knee and elbow joints, hematuria, epistaxis, and circulatory failure.
Indandiones Initial symptoms may range from practically none to pallor, weakness, nausea, severe hematuria, epistaxis and bleeding from mouth and gums. Later symptoms include depression, rapid and labored respiration, accelerated pulse, hemorrhage, bleeding into the thoracic cavity, and circulatory failure.
Inorganics produce a variety of symptoms. Phosphorus is a skin irritant and produces severe burns of skin, mucous membranes, and other tissues with which it comes in contact. Early symptoms include lethargy, restlessness and irritability. Vomiting and diarrhea usually ensue.
Zinc phosphide much less irritating to skin than phosphorus, but it is very irritating to respiratory mucosa. Inhaling dust may produce pulmonary edema. Symptoms include nausea, vomiting, excitement, chills, chest tightness, dyspnea and cough which may progress to pulmonary edema. Severe symptoms include delirium, convulsions, coma, shock from toxic myocardiopathy, jaundice, tetany, anuria, and hemorrhage. Consult treatment for phosphine poisoning in the fumigant section of this manual. Consult also Morgan (1989), Chapter 14, FUMIGANTS, pp. 136-141.
Convulsants:
Sodium Fluoroacetate produces cardiac arrythmia progressing to ventricular fibrillation. Effects on the nervous system are expressed as tonic-clonic convulsions, spasms and rigor.
Strychnine produces tightness and twitching of the muscles, especially
those in the face and neck. Movements may be abrupt. Vomiting may occur.
Generalized convulsions occur within 15 to 30 minutes after ingestion.
They may be clonic at first but quickly become tonic. Convulsions become
progressively severe. Patients remain conscious until respiratory stoppage
produces anoxia and cyanosis. Convulsions may be violent enough to cause
compression fractures of the vertebrae. Legs are adducted and extended,
feet curved inward. Foam gathers at the mouth. Eyes protrude and pupils
dilate. The pulse may be difficult to detect.
Substituted ureas:
Pyraminil produces lassitude, anorexia, constipation and abdominal bloating in the early stages. Later it may produce painful paresthesia with numbness of the extremities and difficulty in walking. Progressive autonomic and peripheral polyneuropathy is characterized by orthostatic hypotension, greatly diminished response to pinprick. Later effects also include diabetes as a result of the effect on cells of the pancreas.
Treatment
Treatments vary with the nature of the agent. Treatment of poisoning by these agents is described in Morgan (1989) pp. 115-130.
RODENTICIDES AND PREDATOR CONTROL AGENTS
| RODENTICIDE TYPE | BRAND | CHEMICAL | SITE-PEST | Morgan (1989) |
| Coumarin | DECON
TOX-HID WARFACIDE |
WARFARIN | RATS & MICE | pp. 115-119 |
| Indandione | RAMIK
GOLD CREST DIPHACINE PROMAR |
DIPHACINONE | RAT, MICE, SQUIRREL | pp. 115-119 |
| Zinc Phosphide | ZP BAIT | ZINC PHOSPHIDE | RAT, MICE | pp. 120 |
| Fluoroacetate | COMPOUND 1080 | SODIUM FLUOROACETATE | COYOTE | pp. 123-124 |
| Botanical | GOPHER GETTER | STRYCHNINE | POCKET GOPHER | pp. 123, 125 |
| Thiourea | ANTU
ANTURAT BANTU KRYSID RAT-TRAK RAT-TU |
-NAPHTHYL THIOUREA | NORWAY RAT | pp. 126-127 |
| Indandione | ROZOL
DRAT QUICK LEPIT SAVIAC |
CHLORPHACINONE | RAT, MICE | pp. 115-119 |
| Coumarin | HAVOC
TALON RATAK VOLAK |
BRODIFACOUM | RAT, MICE | pp. 115-119 |
| Substituted Urea | VACOR (canceled) | PYRAMINIL | RAT, MICE | pp. 126-128 |
| Botanical | DETHDIET
RODINE |
RED SQUILL | pp. 126-128 | |
| Substituted urea | VACOR
PNU HOUSE MOUSE TRACKING POWDER |
PYRAMINIL | RAT, MICE | pp. 128 |
| Coumarin | FUMARIN
FUMASOL KRUMKIL RATFIN RAT-A-WAY TOMARIN |
COUMAFURYL | RAT, MICE | pp. 115-119 |
| Cyanide | M-44
COYOTE GETTER |
SODIUM CYANIDE | COYOTE | pp. 143-145 |
| Vitamin D-related | QUINTOX | CHOLECALCIFEROL | RAT, MICE | pp. 129 |
Toxicology
Effects vary from fungicide type and organ or system it affects. These are usually in the form of dusts, powders and granules for seed and grain, as well as for storage and shipment of these and for mature crop protection.
Chemical Effects
Skin irritants, dermal sensitizers, system manifestations.
Exposure
Skin, mucous membranes
Symptoms/Signs
Skin lesions, scarring, conjunctivitis, keratitis, pyrexia, corneal opacities, hepatomegaly, porphyria, cellulitis, weakness, anorexia, methemoglobinemia
Laboratory
Will vary between different fungicides
Treatment
Vary between hexachlorobenzenes, pentachlorobenzines, diclorean, chloroneb, and thiram
Wash
Lavage and induce vomiting necessary + syrup of ipecac
3-8 gm q.i.d. of cholestyramine will accelerate elimination
Treat porphyria symptoms
Monitor liquids and cardiac functions
FUNGICIDES
| NORTHEAST
(SUBCATEGORIES) |
BRAND |
CHEMICAL |
CROPS |
Morgan (1989) |
| (ETHLENEBISDITHIO CARBAMATE) | DITHANE
MANZATE |
MANEB
MANCOZEB |
PEANUTS, WATERMELONS | pp. 100-101 |
| (BENZONITRILE) | BRAVO | CHLORTHALONIL | MELONS, PEANUTS | pp. 95 |
| (BENZIMDAZOLE) | BENLATE | BENOMYL | MELONS | pp. 112 |
| (PTHALIMIDE) | CAPTAN | CAPTAN | PEACHES | pp. 102 |
| (CHLORINATED HYDROCARBONS) | TERRACHLOR | PCNB | PEANUTS | pp. 17-27 |
| (ORGANOTIN) | DU-TER | TRIPHENYLTIN | PECANS | pp. 108 |
| (BENZIMIDAZOLE) | APL-LUSTER
ARBOTECT |
THIABENDAZOLE | SWEET POTATOES | pp. 113 |
| (CHLOROPHENOXY) | BAYLETON | TRIADIMEFON | WHEAT | pp. 113 |
| SOUTHEAST
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | Morgan (1989) |
| (ETHYLENEBISDITHIOCARBAMATE | DITHANE
MANZATE |
MANCOZEB | WHEAT, SQUASH | pp. 100-101 |
| (BENZONITRILE) | BRAVO | CHLORTHALONIL | SQUASH, WATERMELONS | pp. 112 |
| (DICARBOXIMIDE) | CAPTAN | CAPTAN | PEACHES | pp. 102 |
| (ORGANOTIN) | DU-TER | TRIPHENYLTIN | PECANS | pp. 108 |
| (ELEMENTAL SULFUR) | DUSTING SULFUR | SULFUR | PEACHES | pp. 48-49 |
| TRIZOLE | TILT | PROPICONAZOLE | RICE, WHEAT | None (Call. CIBA-GEIGY
1-800-888-8372) |
| (CHLOROPHENOXY) | BAYLETON | TRIADIMEFON | WHEAT | pp. 113 |
| SOUTH/VALLEY
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | Morgan (1989) |
| (ORGANOTIN) | TORQUE
VENDEX |
FENBUTATINOXIDE | CITRUS | pp. 108-109 |
| (CHLORINATED ISOPTHALIC ACID) | BRAVO | CHLORTHALONIL | CELERY, CAULIFLOWER, CABBAGE, BROCCOLI, MELONS, CUCUMBERS, PEANUTS, ONIONS, TOMATOES, SQUASH, WATERMELONS | pp. 95 |
| (ETHYLENEBISDITHIOCARBAMATE) | DITHANE | MANEB | CELERY, CAULIFLOWER, CABBAGE, BROCCOLI, WATERMELONS, LETTUCE, CUCUMBERS, ONIONS, COLLARDS, MUSTARD GREENS, KALE, SWISS CHARD, KOHLRABI, TOMATOES, TURNIPS, SPINACH, CELERY, MELONS | pp. 100-101 |
| DITHANE,
MANZATE |
MANCOZEB | SQUASH | pp. 100-101 | |
| (BENZIMIDAZOLE) | BENLATE | BENOMYL | CELERY, MELONS, PEACHES, WATERMELONS | pp. 112 |
| (DIMETHYL ANILINE) | RIDOMIL,
APRON |
METALAXYL | MELONS, CUCUMBERS, ONIONS, SORGHUM, SQUASH, SPINACH, WATERMELONS | pp. 113 |
| (CHLORINATED HYDROCARBON) | TERRACLOR | PCNB | PEANUTS | pp. 17-27 |
| (DIOZADIAZOLE) | ROVRAL | IPRODIONE | ONIONS | pp. 112 |
| (DICARBOXIMIDE) | CAPTAN | CAPTAN | PEACHES, SPINACH | pp. 102 |
| (CHLOROPHENOXY) | BAYLETON | TRIADIMEFON | CUCUMBERS, SQUASH, MELONS | pp. 113 |
| (ELEMENTAL SULFUR) | DUSTING SULFUR | SULFUR | PEACHES | pp. 48-49 |
| WEST
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | |
| (CHLORINATED ISOPTHALIC ACID) | BRAVO | CHLORTHALONIL | WATERMELONS, MELONS, ONIONS, POTATOES, PEANUTS | pp. 95 |
| (ETHYLENEBISDITHIOCARBAMATE | DITHANE | MANEB | WATERMELONS, ONIONS, POTATOES, PEANUTS | pp. 100-101 |
| MANZATE | MANCOZEB | ONIONS | pp. 100-101 | |
| (CARBAMATE) | TOPSIN M | THIOPHANATE-METHYL | PEANUTS | pp. 96-101 |
| (BENZIMIDAZOLE | BENLATE | BENOMYL | WATERMELONS, GRAPES | pp. 112 |
| (PTHALIMIDE) | CAPTAN | CAPTAN | GRAPES | pp. 102 |
| (DIMETHYL ANILINE) | RIDOMIL | METALAXYL | MELONS | pp. 113 |
| (ORGANOTIN) | TRIPLE TIN | TRIPHENYLTIN HYDROXIDE | SUGAR BEETS | pp. 108-109 |
| (CHLOROPHENOXY) | BAYLETON | TRIADIMEFON | GRAPES, MELONS, SUGAR BEETS | pp. 113 |
| (CHLORINATED HYDROCARBON) | TERRACHLOR | PENTACHLORO NITROBENZINE | PEANUTS | pp. 17-27 |
Toxicology
Weed killers by selective metabolic impairment unique to plant life. Careless handling in humans affects eyes, skin, mucous membranes.
Chemical Effects
Effects depend on adjuvants (stabilizers, penetrants, safeners, surfactants) or mixed with organophosphates excreted in urine.
Exposure
Occupational/accidental = contact through eyes, skin and mucous membranes; ingestion.
Symptoms/Signs
Skin lesions, scarring, conjunctivitis, keratitis, pyrexia, corneal opacities, hepatomegaly, porphyria, other serious CNS symptoms. Carbamate herbicides will show symptoms similar to the carbamate insecticides.
Laboratory
Generally not available to confirm human absorption; determine recent
exposure from occupation.
NOTE: Some herbicides as chlorophenoxys have lab urine and blood procedures
that are useful for assessing the magnitude of the poisoning.
Treatment
Wash, remove from contact, flush eyes and treat toxicosis. Carbamate herbicides treatment should follow the suggestions given for carbamate insecticides. General treatment guidelines for other herbicides follows:
Gastric lavage may be necessary
Support with IV solutions
Control body temperature with physical means
Pulmonary and cardiac monitoring may be necessary, may need to give oxygen continuously to reduce anoxia
Anticonvulsive therapy may be necessary
For bipyridyl (diquat/paraquat) poisoning administer Bentonite and Fuller's Earth. Consult Morgan (1989), pp. 76-82 for specific patient management. See also table on pp. 84-86 of this manual for trade names of bipyridyl herbicides.
Note: Listen for bowel sounds; ileus may occur
HERBICIDES
| NORTHEAST
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | Morgan (1989) |
| DINITROTOLUIDINE | SURFLAN | ORYZALIN | BLUEBERRIES, PEACHES | pp. 83-88 |
| DINITROTOLUIDINE | TREFLAN | TRIFLURALIN | PEANUTS, WATERMELONS,
SOUTHERN PEAS |
pp. 83-88 |
| DINITROTOLUIDINE | PROWL | PENDIMETHALIN | PEANUTS | pp. 83-88 |
| CHLORO-S-TRIAZINE | PRINCEP | SIMAZINE | BLUEBERRIES | pp. 83-88 |
| BIPIRDYL | GRAMMOXONE | PARAQUAT | BLUEBERRIES, COTTON, SWEET POTATOES | pp. 76-82 |
| ORGANOPHOSPHATE | ROUNDUP | GLYPHOSATE | SWEET POTATOES, BLUE BERRIES, PEACHES, PECANS | pp. 1-11 |
| ALKYL CYCLOHEXENE | POAST | SETHOXYDIM | BLUEBERRIES | pp. 83-88 |
| TRIAZINE | AATREX | ATRAZINE | CORN, SORGHUM | pp. 83-88 |
| TRIAZINE | PRINCEP | SIMAZINE | CORN, ORCHARDS | pp. 83-88 |
| TRIAZINE | PRAMATOL | PROMETON | FENCELINES, EQUIPMENT YARDS, DITCHBANKS | pp. 83-88 |
| TRIAZINE | BLADEX | CYANAZINE | CORN | pp. 83-88 |
| TRIAZINE | SENCOR, | METRIBUZIN | ALFALFA, SOYBEANS | pp. 83-88 |
| TRIAZINE | LEXONE | METRIBUZIN | ALFALFA, SOYBEANS | pp. 83-88 |
| ACETANILIDE | LASSO | ALACHLOR | CORN, PEANUTS | pp. 83-88 |
| ACETAMIDE | DUAL
BICEP |
METOLACHLOR
+ ATRAZINE |
CORN | pp. 83-88 |
| ACETAMIDE | ENID | DIPHENAMID | SORGHUM, SWEET POTATOES | pp. 83-88 |
| PHTHALATES | DACTHAL | DCPA | SOUTHERN PEAS, SWEET POTATOES | pp. 83-88 |
| DICHLOROBENZOIC ACID | AMIBEN | CHLORAMBEN | SWEET POTATOES | pp. 83-88 |
| CHLOROPHENOXY | 2,4-D | 2,4-D | WHEAT, COASTAL BERMUDA | pp. 63-67 |
| BENZOIC ACID DERIVATIVE | BANVEL | DICAMBA | WHEAT, COASTAL BERMUDA | pp. 83-88 |
| SULFONYLUREA | GLEAN | CHLORSULFURON | WHEAT | pp. 83-88 |
| PYRIDAZINONE | SOLICAM | NORFLURAZON | BLUEBERRIES | pp. 83-88 |
| CHLOROPHENOXY | BUTYRAC | 2,4-DB | PEANUTS | pp. 63-67 |
| TRICHLORPICOLINIC ACID | GRAZON | PICLORAM | COASTAL BERMUDA | pp. 83-88 |
| BENZOIC ACID + CHLOROPHENOXY | WEEDMASTER | DICAMBA + 2,4-D | COASTAL BERMUDA | pp. 83-88 |
| SOUTHEAST
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | Morgan (1989) |
| CHLOROPHENOXY | 2,4-D | 2,4-D | WHEAT, COASTAL BERMUDA | pp. 63-67 |
| CHLOROPHENOXY | BUTYRAC | 2,4-DB | PEANUTS | pp. 63-67 |
| SULFONYLUREA | GLEAN | CHLORSULFURON | WHEAT | pp. 83-88 |
| SULFONYLUREA | ALLY | METSULFURON METHYL | WHEAT | pp. 83-88 |
| DICHLORBENZOIC ACID | AMIBEN | CHLORAMBEN | SQUASH | pp. 83-88 |
| DINITRO-TOLUDINE | TREFLAN | TRIFLURALIN | WATERMELONS, PEANUTS | pp. 83-88 |
| DINITROTOLUIDINE | SURFLAN | ORYZALIN | PEACHES, BLUEBERRIES | pp. 83-88 |
| CHLORO-S-TRIAZINE | AATREX | ATRAZINE | CORN, SORGHUM | pp. 83-88 |
| BIPYRIDYL | GRAMMOXONE | PARAQUAT | BLUEBERRIES, ORCHARDS | pp. 76-82 |
| ACETANILIDE | LASSO | ALACHLOR | CORN, SORGHUM | pp. 83-88 |
| ACETAMIDE | DUAL | METOLACHLOR | CORN, SORGHUM, SOYBEANS | pp. 83-88 |
| ACETAMIDE | STAMPEDE | PROPANIL | RICE | pp. 83-88 |
| ORGANOPHOSPHATE | ROUNDUP | GLYPHOSATE | CORN, PEACHES, PECANS, BLUEBERRIES | pp. 1-11 |
| THIADIAZIN | BASAGRAN | BENTAZON | RICE, SOYBEANS | pp. 83-88 |
| CHLORO-S-TRIAZINE | PRINCEP | SIMAZINE | BLUEBERRIES | pp. 83-88 |
| THIOCARBAMATE | BOLERO | THIOBENCARB | RICE | pp. 83-88 |
| PYRIDAZINONE | SOLICAM | NORFLURAZON | BLUEBERRIES | pp. 83-88 |
| ALKYL CYCLOHEXENE DERIVATIVE | POAST | SETHOXYDIM | BLUEBERRIES | pp. 83-88 |
| TRICHLOROPICOLINIC ACID | GRAZON | PICLORAM | COASTAL BERMUDA | pp. 83-88 |
| BENZOIC ACID + CHLOROPHENOXY | WEEDMASTER | DICAMBA + 2,4-D | COASTAL BERMUDA | pp. 63-67 |
| DIPHENYLETHER | BLAZER | ACIFLUOROFEN | SOYBEANS | pp. 83-88 |
| IMIDAZOLINONE | SCEPTER | IMAZAQUIN | SOYBEANS | pp. 83-88 |
| ORGANOPHOSPHATE | PREFAR | BENSULIDE | SQUASH, WATERMELONS | pp. 1-11 |
| SOUTH/VALLEY
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | Morgan (1989) |
| TRIAZINE | AATREX | ATRAZINE | CORN, SORGHUM, SUGARCANE | pp. 83-88 |
| TRIAZINE | PRINCEP | SIMAZINE | NONCROP | pp. 83-88 |
| TRIAZINE | CAPAROL | PROMETRYN | COTTON | pp. 83-88 |
| TRIAZINE | EVIK | AMETRYN | SUGARCANE | pp. 83-88 |
| ORGANOPHOSPHATE | ROUNDUP | GLYPHOSATE | CORN, CITRUS, PEACHES | pp. 1-11 |
| ACETANILIDE | LASSO | ALACHLOR | CORN, PEANUTS | pp. 83-88 |
| DIMETHYLUREA | KARMEX | DIURON | CITRUS, SUGARCANE | pp. 83-88 |
| URACIL | HYVAR | BROMACIL | CITRUS | pp. 83-88 |
| DINITROANALINE | TREFLAN | CAULIFLOWER, COTTON, TURNIPS, SUGARCANE, GRAPES | pp. 83-88 | |
| CHLOROPHENOXY | BUTYRAC | 2,4-DB | PEANUTS | pp. 83-88 |
| DINITROTOLUIDINE | SURFLAN | ORYZALIN | PEACHES | pp. 83-88 |
| SULFONAMIDE | BETASAN
PREFAR |
BENSULIDE | LETTUCE | pp. 83-88 |
| BENZOIC ACID | BANVEL | DICAMBA | SUGARCANE | pp. 83-88 |
| WEST
(SUBCATEGORIES) |
BRAND | CHEMICAL | CROPS | Morgan (1989) |
| SULFONYLUREA | ALLY | METSULFURON METHYL | WHEAT | pp. 83-88 |
| SULFONYLUREA | GLEAN | CHLORSULFURON | WHEAT | pp. 83-88 |
| DINITROANILINE | TREFLAN | TRIFLURALIN | COTTON, WATERMELONS, GRAPES, POTATOES | pp. 83-88 |
| DINITROANILINE | SURFLAN | ORYZALIN | GRAPES | pp. 83-88 |
| CYCLOHEXONES | POAST | SETHOXYDIM | SUGAR BEETS | pp. 83-88 |
| BIPYRIDYL | GRAMOXONE | PARAQUAT | POTATOES | pp. 76-82 |
| CHLOROPHENOXY | 2,4-D | 2,4-D | BARLEY, WHEAT | pp. 63-67 |
| ORGANOPHOSPHATE | DEF | DEF | COTTON | pp. 1-11 |
| TRIAZINE | SENCOR | METRIBUZIN | POTATOES | pp. 83-88 |
| DIPHENYL ETHER | GOAL | ONIONS | pp. 83-88 | |
| BENZENE SULFONAMID | PREFAR | BENSULIDE | ONIONS | pp. 83-88 |
| BENZONITRILE | BUCTRIL | BOMOXYNIL | SEED SORGHUM | pp. 83-88 |
| METHYL SULFONATE | NORTRON | ETHOFUMESATE | SUGAR BEETS | pp. 83-88 |
| ORGANOPHOSPHATE | PREFAR | BENSULIDE | MELONS, ONIONS | pp. 1-11 |
| CARBANILATE DERIVATIVE | BETANAL | PENMEDIPHAM | SUGAR BEETS | pp. 83-88 |
| THIOCARBAMATE | EPTAM
GENEP |
EPTC | ALFALFA, SUGAR BEETS | pp. 83-88 |
Toxicology
Metal and nonmetal physical properties; has reversible combination effect on tissue proteins and enzymes; competes with phosphates; causes injury to never cells, blood vessels, liver, kidney and other tissues.
Chemical Effects
Will depend on its biochemical transformation mechanisms: (vascular dilation)
Exposure
Mucous membrane absorption, dermal, ingestion.
Symptoms/Signs
Acute: within 1 hour, garlic odor in breath and feces; mouth pharynx and esophagus inflammation; burning abdominal pain; thirst; vomiting, diarrhea. Renal injury, CNS disorders, cardiovascular and liver damage, anemia, leukopenia, thrombocytopenia, circulatory failure, death.
Chronic
Dermal signs more prominent; hyperkeratosis; hyperpigmentation; dermatitis; subcutaneous edema of face, edema of eyelids and ankles; loss of nails or hair; stomatitis; weight loss; peripheral neuropathy; liver injury; EKG anomalies; anemia; skin cancer; lung cancer; rarely encephalopathy.
Laboratory
24 hour urinary measurement;
GUTZEIT Test
REINSOIT Test
Treatment
Wash skin and hair with copious amount of soap and water.
Gastric lavage and intubation may be necessary
Hydrate with IV fluid if necessary
Cardiac monitoring is important
5. Dimercaprol (BAL) then at 3 mg/kg 1/4 hrs over 3-10 days to 3 mg/kg
1/12 hrs and then after the GI tract is reasonably free of arsenic, oral
administration of D-Penicillamine should probably replace Bal therapy in
persons not allergic to penicillin.
ARSENICALS
INORGANIC ARSENICALS
ORGANIC ARSENICAL HERBICIDES
Arsenicals are sometimes used in combination with other pesticides because of their binding effect. They are mostly used as defoliants, herbicides and insecticides in the form of powders or solutions. Commonly, they have been used in cotton, potatoes, tomatoes, as wood preservatives and as ant killers. Arsenic acid ceased to have application in cotton after cotton processors were forced to dispose of waste water as toxic waste due to arsenic residues from treated cotton. Arsenicals have very few applications outside of ornamental home and garden treatments to control crabgrass. Farm workers from Central America and South America may have high residues of arsenic in their bodies due to extended exposure in countries where arsenicals still have wide use.
Treatment for the arsenical insecticides and herbicides is described in Morgan (1989) pp. 54-62.
Toxicologists continue the common practice of grouping together under the category of fumigants several groups of compounds that have little in common except toxicity to one or more pests and relatively high vapor pressures. Some of the compounds are gases at room temperature. Others are liquids or solids. Their activity against pests depends on their vapors.
Toxicology
The general toxicology of the fumigants varies with the type of compound. The toxicity of many of the compounds is a physical property and follows Ferguson's principle, being lethal at thermodynamic activities between 0.1 and 1.0. Others are volatile chemical poisons and are active at thermodynamic levels far below 0.1.
Chemical Effects
Effects vary with chemical nature of the fumigant from none to strong irritation of eyes, skin, and upper respiratory tract.
Exposure
Skin, respiratory, mucous membranes, oral.
Symptoms/Signs
Symptoms vary with product. A few symptoms include headache, nausea, muscle aches, irritation of eyes, nasal and pharyngeal passages, pulmonary edema, lassitude, mental disorientation, convulsions, hemolysis, cyanosis, and coma.
Laboratory
Will vary between fumigants.
Treatment
Eleven general steps in treating poisoning by fumigants are listed in Morgan (1989), pp. 138-141. They are listed as follows:
1. FLUSH contaminating fumigants from the skin and eyes with copious amounts of water or saline for at least 15 minutes. some fumigants are corrosive to the cornea and may cause BLINDNESS. Specialized medical treatment should be obtained promptly following removal of toxicant by copious flushing with clean water. Skin contamination may cause BLISTERING and deep chemical burns. Absorption of some fumigants across the skin may be sufficient to cause systemic poisoning in the absence of fumigant inhalation. For all these reasons, decontamination of eyes and skin must be IMMEDIATE and THOROUGH.
2. REMOVE TO FRESH AIR IMMEDIATELY: Remove victims of fumigant inhalation to FRESH AIR immediately. Even though initial symptoms and signs are mild, keep the victim quiet, in a semi reclining position Minimum physical activity limits the likelihood of pulmonary edema.
3. IF VICTIM IS NOT BREATHING, RESUSCITATE: If victim is not breathing, clear the airway of secretions and RESUSCITATE, with positive pressure oxygen apparatus. If this is not available, use chest compression to sustain respiration. If victim is pulseless, employ cardiac resuscitation.
4. IF PULMONARY EDEMA IS EVIDENT, there are several measures available to sustain life. Medical judgement must be relied upon, however, in the management of each case. The following procedures are recommended (Morgan, 1989, pp. 139):
(AUTHOR'S NOTE: CHECK FUMIGANT LABELS FOR ANY CONTRAINDICATIONS TO DRUGS.)
Put the victim in a SITTING position with a backrest.
Use intermittent and/or continuous positive pressure OXYGEN to relieve hypoxemia. (Do not give oxygen at greater concentrations or longer periods than necessary, because it may exaggerate fumigant injury to lung tissue. Monitor arterial pO2)
Slowly administer FUROSEMIDE, 40 mg, or SODIUM ETHACRYNATE, 50 mg, intravenously, to reduce venous load by inducing diuresis. Consult package insert for additional directions and warnings.
Administer MORPHINE in small doses (5-10 mg), slowly, intravenously, to allay anxiety and promote deeper respiratory excursions.
Administer AMINOPHYLLINE (0.25-0.50 gm) slowly, intravenously. Consult package insert.
Digitalization may be considered, but there is a serious risk of arrhythmias in an anoxic and toxic myocardium.
TRACHEOSTOMY may be necessary in some cases to facilitate aspiration of large amounts of pulmonary edema fluid.
Epinephrine, atropine, and expectorants are generally not helpful, and may complicate treatment.
5. Combat SHOCK by placing victim in the Trendelenburg position and administering plasma, whole blood, and/or electrolyte and glucose solutions intravenously, with great care, to avoid pulmonary edema. Central venous pressure should be monitored continuously. Vasopressor amines must be given with great caution, because of the irritability of the myocardium.
6. Control CONVULSIONS. Seizures are most likely to occur in poisonings by methyl bromide, hydrogen cyanide, acrylonitrile, phosphine, and carbon disulfide.
Establish pulmonary gas exchange at the best possible level by administering OXYGEN by continuous positive pressure ventilation.
In poisoning by CYANIDE and ACRYLONITRILE, proceed directly with ANTIDOTAL therapy (see Morgan, 1989, pp.143-144).
Control convulsions caused by other agents with careful IV,, injection of DIAZEPAM, 5-10 mg in adults and children over 12 years, 0.25-0.40 mg/kg in children under 12 years. (See Morgan, 1989, Chapter 3, TREATMENT, Section 4, p. 21.) Repeat dosage in g 4-6 hours if necessary. CAUTION: Be prepared to maintain pulmonary ventilation mechanically, and to manage hypotension and cardiac arrhythmias. Alternative or supplemental anticonvulsive therapy is discussed in the reference cited.
In methyl bromide poisoning, it may be necessary to give benzodiazepines or barbiturates orally for days or weeks after the poisoning to control involuntary motor activity. Consult s package inserts for appropriate dosages.
7. If a FUMIGANT LIQUID OR SOLID has been INGESTED less than several hours prior to treatment, quantities remaining in the stomach must be removed as effectively as possible by gastric intubation, aspiration, and lavage, after all possible precautions have been taken to protect the respiratory tract from aspirated gastric contents.
Put in place a cuffed ENDOTRACHEAL TUBE prior to gastric intubation. Administer OXYGEN, using a mechanical ventilator if respiration is depressed.
Lavage the stomach with a slurry of ACTIVATED CHARCOAL in saline or water. Leave a volume of the slurry in the stomach with an appropriate dose of sorbitol as cathartic (for dosages, see Morgan, 1989, Chapter 1, TREATMENT, Section 6, pp. 8).
If treatment is delayed and if the patient remains fully alert, administer activated charcoal and sorbitol orally. For dosage, (see Morgan, 1989, Chapter 1, TREATMENT, Section 6, pp. 8). Repeated administration of charcoal at half or more the initial dosage every 2-4 hours may be beneficial.
Do not give vegetable or animal fats or oils, which enhance gastrointestinal absorption of many of the fumigant compounds.
8. Intravenous infusions of GLUCOSE are valuable in limiting the hepatotoxicity of many substances. Monitor central venous pres sure to avoid precipitating, or aggravating, pulmonary edema by fluid overload. The victim should be watched closely for indications of delayed or recurrent pulmonary edema, and for bronchopneumonia. Fluid balance should be monitored, and urine sediment should be checked regularly for indications of tubular injury. Measure serum alkaline phosphatase, LDH, ALT, AST, and bilirubin to assess liver injury.
9. HEMOPERFUSION OVER ACTIVATED CHARCOAL has been used in managing a case of carbon tetrachloride poisoning with apparent success. An extraction efficiency of about 80% was demonstrated for the system employed (Schwarzbeck, A. and Kosters, W., Arch. Toxicol., 35:207-211, 1976). It is possible that other fumigant compounds would be effectively removed from blood by this method.
10. EXTRACORPOREAL HEMODIALYSIS may be needed to regulate extracellular fluid composition if renal failure supervenes. It is probably not very effective in removing lipophilic fumigant compounds from blood, but is, of course, effective in controlling extracellular fluid composition if renal failure occurs.
11. Certain SPECIFIC MEASURES are recommended in Morgan (1989) for poisonings by particular fumigants (naphthalene, methyl bromide, carbon tetrachloride, hydrogen cyanide). Refer to Morgan page reference in fumigant list.
Morgan provides insufficient or no information on dazomet (Basamid®),
phosphine (Phostoxin®, Aluminum phosphide, Zinc phosphide), and sulfuryl
fluoride. Where information is unavailable from manufacturer, the emergency
telephone number is provided in the fumigant list in the reference column.
Toxicology
Intensive prolonged inhalation exposure, ingestion, or prolonged dermal contact with treated fabric may cause hemolysis in persons deficient in glucose-6-phosphate dehydrogenase. Secondary renal tubular damage may ensue from excretion of the naphthol, other naphthalene metabolites, and products of hemolysis.
Naphthalene produces selective but reversible damage to the nonciliated bronchiolar epithelium. If ingested, Naphthalene may produce local irritation of the gastrointestinal tract.
Convulsions and coma may occur, particularly in children. In infants, high levels of hemoglobin, methemoglobin, and bilirubin in the plasma may lead to encephalopathy (kernicterus). Some individuals exhibit dermal sensitivity to naphthalene.
Chemical
Naphthalene is a solid white hydrocarbon, the main constituent of moth balls. It is packaged in ball, flake, or cake form as a moth repellent. It sublimes slowly. It has a sharp, pungent odor that is irritating to eyes and upper respiratory passages.
Signs/Symptoms
High concentrations produce headache, dizziness, nausea and vomiting. Acute poisoning also may produce ptosis and clear red secretions around the eye. Chronic toxicity produces decrease in body weight and loss of appetite.
Laboratory
Chromatography: Naphthalene is converted mainly to alpha naphthol in the body and promptly excreted in conjugated form in the urine. Alpha naphthol can be measured by gas chromatography.
Treatment
Morgan (1989) provides the following specific suggestions for treatment of naphthalene toxicosis on pp. 141.
A. NAPHTHALENE toxicosis caused by vapor inhalation can usually be managed simply by removing the individual to fresh air. Skin contamination should be removed promptly by washing with soap and water. Eye contamination should be removed by flushing with copious amounts of clean water. Irritation may be severe, and if it persists, should receive medical attention.
If solid naphthalene has been INGESTED and retained less than several hours prior to treatment, and if the patient is fully alert, the stomach should be emptied by administration of Syrup of Ipecac, followed by several glasses of water. Dosage for adults and children over 12 years: 30 ml; dosage for children under 12 years: 15 ml. When vomiting subsides, give activated charcoal and sorbitol (see Morgan, 1989, Chapter 1, TREATMENT, Section 6, p. 8). If the patient is obtunded or excited, do not give Ipecac, but take steps to protect the airway, then aspirate and lavage the stomach with a slurry of activated charcoal. Leave charcoal and sorbitol in the stomach before withdrawing the tube (see above reference). Repeated administration of charcoal every 2-4 hours may be beneficial.
If treatment is delayed more than several hours, administer as much activated charcoal orally as the patient will tolerate. Include sorbitol in the charcoal slurry unless diarrhea has already commenced.
Examine the plasma for evidence of hemolysis: a reddish-brown tinge. Examine the blood smear for "ghosts" and Heinz bodies. If present, monitor red blood cell count and hematocrit for anemia, urine for protein and cells. Measure direct- and indirect-reacting bilirubin in the plasma. Monitor fluid balance and blood electrolytes. If possible, monitor urinary excretion of naphthol to assess severity of poisoning.
Toxicology
Liver injury and tremor may occur following ingestion of large amounts. Although accidental ingestions especially by children, have been fairly common, symptomatic human poisonings have been rare. Other stereoisomers of dichlorobenzene are more toxic than the para-isomer. Some nervous system effects have been observed through excitation of axonic transmission. Ingestion has showed slight increase in weight of liver and kidneys and a slight focal necrosis and cirrhosis of the liver. However, evidence shows paradichlorobenzene partitions into fat at 10 to 40 times the rate it concentrates in liver or kidneys.
Liver necrosis observed with paradichlorobenzene is proportional to the degree of covalent binding of active metabolites to liver proteins. Paradichlorobenzene has induced some porphyria. It also has produced increased urinary coproporphyrin excretion.
Studies of chronic effects have showed mutagenicity and carcinogenicity in experimental animals. These included renal tubular cell adenocarcinomas, hepatocellular carcinomas, and pheochromocytomas.
Chemical Effects
Paradichlorobenzene is the active principle in commercial "moth crystals" and many home garden formulations designed to control wood boring insects. It is solid at room temperature, and is now widely used as a moth repellant, air freshener, and deodorizer in homes and in public facilities. It is commonly placed in public rest rooms in the form of "toilet cakes" or "urinal cookies." The vapor is only mildly irritating to the nose and eyes.
Exposure
Respiratory, dermal, oral
Symptoms/Signs
Symptoms depend upon the amount and route of exposure. They include mild irritation to nose and eyes, headache, weakness, muscle twitches, tremors, loss of equilibrium, horizonal and vertical nystagmus, and rapid labored breathing, and coma. Reversible eye ground changes and systemic changes have been observed.
Laboratory
Chromatography: Paradichlorobenzene is metabolized mainly to 2,5-dichlorophenol, which is conjugated and excreted in the urine. This product can be measured chromatographically.
Treatment
Treatment for paradichlorobenzene toxicosis is outlined above in Morgan's
general treatments for fumigants and may be found in the Morgan (1989)
manual, pp. 137-143.
Toxicology
Carbon tetrachloride is toxic to central nervous system and liver. It less toxic than chloroform as a central nervous system depressant, but is much more severely hepatotoxic, particularly following ingestion. Liver cell damage is apparently due to a free radical generated in the process of initial dechlorination. Kidney injury also occurs; sometimes this is exaggerated by jaundice. Cardiac arrhythmias, progressing to fibrillation, may follow inhalation of high concentrations of carbon tetrachloride or ingestion of the liquid.
Carbon tetrachloride impairs the NADPH-dependent oxidative enzymes in liver microsomes by causing irreversible damage to cytochrome P-450. It does not act as a competitive inhibitor. In the liver, carbon tetrachloride produces elevated levels of glutamic-oxaloacetic transaminase and aldolase (commonly used in following the clinical course of human patients poisoned by the compound).
Centrolobular necrosis of the liver is the lesion most characteristic of poisoning by carbontetrachloride. The necrosis progresses cell by cell. Electron microscopy reveals vesiculation of the rough endoplasmic reticulum, formation of clumps of tangled smooth membranes and vacuolization of the Golgi apparatus. It also reveals loss of polysomes and accumulation of fat.
Definite renal tubular lesion, including tubular necrosis and deposition of calcium have been observed regularly. Mitochondira and not endoplasmic reticulum appears to be the primary subcellular site of carbon tetrachloride toxicity in the kidney.
Chemical effects
Carbon tetrachloride is a colorless liquid with a sweetish odor. It is nonflammable and noncorrosive. It is inert generally but is deomposed by water at high temperatures. It is not particularly irritating at low concentrations, but in higher concentrations gives a suffocating sensation.
Exposure
Respiratory, oral, dermal. Carbon tetrachloride is readily absorbed by tissues including the linings of the respiratory and digestive tracts. It also will pass through the skin.
Symptoms/Signs
Symptoms of Carbon tetrachloride poisoning may include giddiness, sleepiness, and some dizziness. In the case of ingestion, there may be some increase in peristalsis. There is respiratory excretion (indicated by odor oof the breath as well as by chemical analysis. Skin contamination may produce erythemia as well as signs of carbon tetrachloride respiratory excretion. Alcohol consumption increases the toxic effects of carbon tetrachloride. Acute symptoms include kidney and liver failure, narcosis, and gastroenteritis
Laboratory
Many halocarbons, including carbon tetrachloride, can be measured in blood by gas chromatographic methods, some using head space techniques. Some, including, carbon tetrachloride can be measured in the expired air as well.
Treatment
Morgan (1989, pp. 142) outlines specific treatment for carbon tetrachloride poisoning CARBON TETRACHLORIDE poisoning, several treatment measures have been suggested to limit the severity of hepatic necrosis. Neither effectiveness nor safety of any of these measures has been established.
Inhalation of oxygen at one or two atmospheres for 2 hours twice daily may have some value.
Oral administration of tocopherol (vitamin E) in oral doses of several hundred milligrams per day has been suggested on grounds of its action as a free radical scavenger.
Oral administration of N-acetyl cysteine (Mucomyst) may be worthwhile as a means of reducing free radical injury. Dilute the proprietary 20% product 1:3 in soda pop, and give about 3 ml/kg body weight of the diluted solution as a loading dose. G*e half of this dosage every 4 hours after the loading dose for a total of 17 doses. (This dosage schedule is used for acetaminophen poisonings.) Administration via duodenal tube may be necessary in a few patients who cannot tolerate Mucomyst.
Hemoperfusion over activated charcoal should be considered. It was apparently effective in one carbon tetrachloride poisoning. See Schwarzbeck, A. and Kosters, W. Arch. Toxicol., 35:207-211,1976.
Toxicology
Carbon disulfide vapor is only moderately irritating to upper respiratory membranes, but it has an offensive "rotten cabbage" odor. Acute toxicity is due chiefly to effects on the central nervous system. Long-term occupational exposures have been shown to accelerate atherosclerosis, leading to ischemic encephalopathy, myocardiopathy, and gastrointestinal dysfunction. Toxic damage to the liver and kidneys may result in severe functional deficits of these organs.
Chemical Effects
Impurities give carbondisulfide a foul smell. However, in the pure form, it has a sweetish odor.
Exposure
Respiratory, oral, dermal. Carbon disulfide is readily absorbed from the respiratory and gastrointestinal tracts.
Symptoms/Signs
Symptoms demonstrate the effects of carbon disulfide on the nervous system. They include drowsiness, motor weakness, flaccid parylisis, nerve tenderness, staggering and sumbling as though drunk, extreme thirstiness, loss of appetite, behavioral changes, rigidity and tremor (parkinsonism), choreatic movments. Inhalation of high concentrations for short periods has caused headache, dizziness, nausea, hallucinations, delirium, progressive paralysis and death from respiratory failure. More prolonged exposure to lesser amounts has lead to blindness, deafness, paresthesia, painful neuropathy, and paralysis.
Laboratory
Carbon disulfide can be measured in urine by gas chromatography, but the test is not generally available. A qualitative test for carbon disulfide metabolites in urine (based on their reducing properties) is used for monitoring occupational exposure (Djuric D., N. Serducki, and I. Burkes. Iodine-azide test on urine of persons exposed to carbon disulfide. Brit. J. Indus. Med., 22:321-3, 1965).
Treatment
Morgan (1989, pp. 143) suggests the following treatments for carbon disulfide poisoning: Mild poisonings by CARBON DISULFIDE inhalation may be managed best by no more than careful observation, even though sensory hallucinations, delirium, and behavioral aberrations can be alarming. Severe poisonings may require specific measures:
A. If manic behavior threatens the safety of the victim, DIAZEPAM, 5-10 mg in adults, 0.2-0.4 mg/kg in children, administered slowly, intravenously, may be helpful as a tranquilizer. Give as much as is necessary to achieve sedation. Do not give catecholamine-releasing agents such as reserpine and amphetamines.
B. In severe poisonings by carbon disulfide, pyridoxine hydrochloride (vitamin B6) may have some antidotal action against the neurotoxic effects. Its value is theoretical; neither effectiveness nor safety has been tested in carbon disulfide poisonings. The usual dosage in other poisonings (ISONIAZID) has been 5 gm in a 10% solution, given slowly intravenously, or included in a one liter intravenous solution of 5% glucose. When the victim can swallow, pyridoxine hydrochloride can be given orally in daily doses as high as 25 mg/kg body weight. There is probably little value, and possibly some hazard, in extending the treatment beyond one or two weeks.
Toxicology
Hydrogen cyanide gas causes poisoning by inactivating cytochrome oxidase, the final enzyme essential to mammalian cellular respiration. The cells of the brain appear to be the most vulnerable to cyanide action. Similar color of the retinal arteries and veins may be a useful sign of cyanide poisoning; it is due to failure of reduction of hemoglobin as blood perfuses poisoned tissues. Cyanide poisoning does not produce cyanosis but leaves the venous blood fully oxygenated and the patient pinker than normal.
The cyanide ion has essentially the same toxicity, regardless of the route by which it is absorbed. Whether the route of exposure was oral or respiratory, victims experienced an almost instantaneous collapse and cessation of respiration. After poisoning, the heart may continue for some time after respiration stops. While respiration is active, the venous bllod reains oxygenated and the patient's color florid. In fact, this condition may persist if death is sudden.
Persons other than applicators who were poisoned accentally because they remained in a fumigated space or accidentally triggered an M-44 Coyote Getter, usually bllundered unknowingly into a contaminated environment.
Chemical Effects
The toxicity of hydrogen cyanide, simple cyanide salts, and other compounds such as cyangen and acrylonitrile depends upon their ability to yield the cyanide ion. Hydrogen cyanide has the characteristic sharp smell of bitter almonds. The ability of acrylonitrile to destroy human epidermis is most likely a local effect of the unmetabolized molecule. It is a curious fact that blood levels in fatal cyanide poisoning usually are higher following ingestion (3.2-160 ppm) than following inhalation (0.5-15) ppm.
Acrylonitrile is biotransformed in the body to hydrogen cyanide. Toxicity and mechanisms of poisoning are essentially the same as have been described for cyanide, except that acrylonitrile is irritating to the eyes and to the upper respiratory tract.
Exposure - Respiratory, oral, dermal.
Symptoms/Signs
Unconsciousness and death may occur immediately following inhalation of a high cyanide concentration, respiratory paralysis being the principal mechanism. Lesser exposures cause a constriction and numbness in the throat, stiffness of the jaw, salivation, nausea, vomiting, dizziness, and apprehension. Worsening of the poisoning is manifest as violent tonic or clonic convulsions. Trismus and opisthotonos occur. Paralysis follows seizure activity. Incontinence is characteristic. The skin remains pink. Fixed, dilated pupils, bradycardia, and irregular gasping respiration (or apnea) are typical of profound poisoning. The heart often continues to beat after breathing has stopped. A bitter almond odor to the breath or vomitus may be a clue to poisoning, but not all individuals are able to detect this odor.
Laboratory
Cyanide ion from cyanide itself or acrylonitrile can be measured in whole blood and urine by an ion-specific electrode or by colorimetry. The upper limit in whole blood among nonexposed nonsmokers is about 0.02 mg per liter; it is 0.04 mg per liter in smokers. Symptoms may appear at levels above 0.10 mg per liter. Urine cyanide is usually less than 0.30 mg per liter in nonsmokers, but as much as 0.80 mg per liter in smokers. Thiocyanate, the metabolite of cyanide, can also be measured in blood and urine. It is usually present in plasma at levels less than 4 mg per liter in non-smokers, but up to 12 mg per liter in smokers Urine thiocyanate is usually less than 4 mg per liter in non | smokers, but may be as high as 17 mg per liter in smokers.
Treatment
Morgan (1989, pp. 143-145) makes the following specific suggestions for treating cyanide poisoning:
Poisonings by HYDROGEN CYANIDE and ACRYLONITRILE gases or liquids
are treated essentially the same as poisoning by cyanide salts. Because
cyanide is so promptly absorbed following ingestion, treatment should commence
with PROMPT ADMINISTRATION OF ANTIDOTES, deferring gastric evacuation
(in ingestion poisonings) until antidotes have been administered.

Morgan (1989) states, "Although various cobalt salts, chelates,
and organic combinations have shown some primise as antidotes to cyanide,they
are not generally available. None have been show to surpass the nitrite-thiosulfate
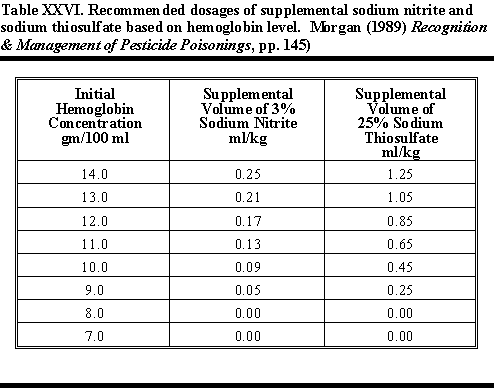
regimen in effectiveness." Table XXVI shows Morgan's (1989) table
of recommended dosages of supplemental soidum nitrite and sodium thiosulfate
based on hemoglobin level.
Administer OXYGEN continuously. If respiration fails, maintain pulmonary ventilation mechanically.
Administer AMYL NITRITE (perles) by inhalation for 15-30 seconds of every minute, while a fresh solution of 3% sodium nitrite is being prepared.
As soon as solution is available, inject intravenously 10 ml of 3% SODIUM NITRITE solution over a 2-4 minute interval, keeping the needle in place. CAUTION: MONITOR PULSE and BLOOD PRESSURE during administration of amyl nitrite and sodium nitrite. If systolic blood pressure falls below 80 mm Hg, slow or stop nitrite administration until blood pressure recovers.
Follow sodium nitrite injection with an infusion of 50 ml of 25% aqueous solution of SODIUM THIOSULFATE administered over a 10-minute period. Initial adult dose should not exceed 12.5 gm.
If symptoms persist or recur, treatment by sodium ni trite and sodium thiosulfate should be REPEATED AT HALF THE DOSAGES listed in paragraphs c and d.
Measure hemoglobin and methemoglobin in blood. If more than 50% of total hemoglobin has been converted to methemoglobin, BLOOD TRANSFUSION or exchange transfusion should be considered, because conversion back to normal hemoglobin proceeds slowly.
B If the victim is a CHILD:
Toxicology
Chloropicrin is severely irritating to the upper respiratory tract, eyes, and skin. Inhalation of an irritant concentration sometimes leads to vomiting. Ingestion could be expected to cause a corrosive gastroenteritis. Chloropicrin is lethal in 10 minutes at 2000 mg/m3. It is intolerable at 50 mg/m3. It causes lacrimation and eye irritation at 2 mg/m3. The odor can detected at 7.3 mg/m3. The threshold limit value is 0.7 mg/m3.
Chemical Effects
Chloropicrin is a colorless, slightly oilliquid with an intense odor. It is sold as a grain fumigant and soil fumigant. It is also mixed with other fumigants to increase effectiveness or as a warning agent in the case of 98% methyl bromide formulations. Chloropicrin was employed as a chemical warfare agent during World War I. Even then, its main value was its irritating properties.
Exposure
Respiratory, mucous membranes, dermal, oral.
Symptoms/Signs
Common symptoms of chloropicrin exposure are eye, nose and throat irritation and inflamation; also lacrimation, coughing, sore throat, and vomiting. Others include vertigo, headache, nausea, and fatigue. Some persons exposed to chlorpicrin reported dizziness, drowsiness, wheezing, blurred vision, skin irritation, headache, and a bad taste in the mouth.
Laboratory
There are few laboratory procedures to confirm poisoning by chloropicrin.
Treatment
Treatment is symptomatic. Morgan (1989, pp. 138-141) indicates the following: Maintain pO2 above 60 mm Hg by instituting the following measures stepise as needed (see 11 steps above).
Toxicology
The onset of respiratory distress may be delayed 4-12 hours after exposure. Methyl bromide sometimes induces pulmonary edema, hemorrhage, or a confluent pneumonia. It is a central nervous system depressant and has prounounced effects on the CNS. It also may cause convulsions.
Chemical Effects
Methyl bromide is colorless and nearly odorless, but is severely irritating to the lower respiratory tract. If liquid methyl bromide contacts the skin, severe burning, itching, and blister formation occurs. Skin necrosis may be deep and extensive.
Exposure
Respiratory, dermal, eye.
Symptoms/Signs
Early symptoms of acute poisoning include headache, dizziness, nausea, vomiting, tremor, and ataxia. Repeated prolonged exposures in some cases have led to a long-lasting syndrome of ataxia, incoordination, muscle weakness and areflexia. One case of recurrent myoclonic seizures has been reported which required treatment for five years following methyl bromide exposure.
Laboratory
Methyl bromide yields inorganic bromide in the body; the anion is slowly excreted in the urine (half-life in the body is about 12 days). The serum from persons having no exceptional exposure to bromide usually contains less than 1 mg bromide ion per 100 ml. The possible contributions of medicinal bromides to elevated blood content and urinary excretion must be considered, but if methyl bromide is the exclusive source, serum bromide exceeding 5 mg per 100 ml probably means some absorption, and 15 mg per 100 ml is consistent with symptoms of acute poisoning. Inorganic bromide is considerably less toxic than methyl bromide; serum concentrations in excess of 150 mg per 100 ml occur commonly in persons taking inorganic bromide medications. In some European countries, blood bromide concentrations are monitored routinely in workers exposed to methyl bromide. Blood levels over 3 mg per 100 ml are considered a warning that personal protective measures must be improved. A bromide concentration over 5 mg per 100 ml requires that the worker be removed from the fumigant contaminated environment until blood concentrations decline to less than 3 mg per 100 ml.
Treatment
Morgan (1989, pp. 140-142) states: "In methyl bromide poisoning, it may be necessary to give benzodiazepines or barbiturates orally for days or weeks after the poisoning to control involuntary motor activity. Consult package inserts for appropriate dosages."
"If given very soon after life-threatening exposure to METHYL BROMIDE there may be some theoretical value in administering DIMERCAPROL (BAL) in vegetable oil intramuscularly. For adults, give 3-5 mg/kg q6h for 4 to 6 doses. Neither the effectiveness nor the safety of this treatment has been tested in methyl bromide poisoning. CAUTION: DIMERCAPROL may cause troublesome side effects (hypertension, tachycardia, nausea, headache, paresthesia, pain, lacrimation, sweating, anxiety, and restlessness). Although usually not so severe as to preclude treatment, these effects may require antihistamine therapy."
Toxicology
Dichloropropene and dichloropropane are strongly irritating to the skin, eyes, and respiratory tract. Bronchospasm may result from inhalation of high concentrations. Liver, kidney, and cardiac toxicity is probably similar to that produced by carbon tetrachloride. It also procudes nontumorigenic lesions of the nasal mucosa and changes in in the morphology of renal and hepatic tissues.
Chronic effects include mutagenicity but not carcinogenicity. It is weakly mutagenic on liver microsomes. Formulations of 1,3-dichloropropene form mutagenic oxidation product. Chronic toxicity and oncogenicity studies have generally produced negative results.
Chemical Effects
Dichloropropene is a colorless to straw colored liquid. It is severely irritating to skin, eyes, and upper respiratory tract. It also is extremely flammable with a flash point of 92 F (25 C).
Exposure
Respiratory, eyes, skin.
Symptoms/Signs
Exposure to low concentrations prouces fatigue, desensitization or loss of the sense of smell, headache, and chest discomfort. It may not produce immediate symptoms of strong irritation to eyes, skin or respiratory passages.
Exposure to higher concentrations produces strong irritation of mucuous membranes, chest discomfort, headache, weakness, unconsciousness.
Laboratory
Dichloropropene and dichloropropane like many halocarbons can be measured in blood by gas chromatographic methods, some using head space techniques. Like some other halocarbons, it also can be measured in the expired air.
Treatment
Morgan (1989) provides treatment guidlines in 11 steps on pages 138-141.
Labels provide the following precautionary note to physicians:
"Because rapid absorption may occur through lungs if product is aspirated and cause systemic effects, the decision to induce vomiting or not should be made by a physician. If lavage is performed, endotracheal and/or esophageal control is suggested. Danger from lung aspiration must be weighted against toxicity when considering emptying the stomach."
Toxicology
Dibromochloropropane is irritating to skin, eyes, and the respiratory tract. Exposure produces slight to mderate CNS depression Eye damage has resulted from repeated exposure to the vapors. Liver and kidney damage are prominent features of acute poisoning.
Chronic exposure to relatively low concentrations has led to permanent sterility of workers in a manufacturing plant, by causing diffuse necrosis of seminiferous tubule cells. Because it is much less odiferous than ethylene dibromide, exposure of workers to toxic concentrations of DBCP is more likely.
Men with chronic exposure to DBCP were mildly to severely oligospermic with some totally azoospermic. An analysis of semen from DBCP-exposed workers showed positive correlation between length of exposure to DBCP and the extent of reduction in sperm production. DBCP-exposed men also produce serum levels of follicle-stimulating hormone (FSH) and leuteinizing hormone (LH).
Other chronic effects include gastric squamous carcinoma, papilloma, and tumors of the stomach, lung, and nasal cavity.
Chemical Effects
Dibromochloropropane produces respiratory irritation at vapor concentrations of 60 ppm or higher.
Exposure
Respiratory, skin, eye
Symptoms - Symptoms include headache, nausea, vomiting, ataxia, and slurred speech.
Symptoms of human poisoning also include severe continuous cramping, epigastric and right upper quadrant pain, fever, anorexia, nausea, vomiting and diarrhea.
Laboratory
Dibromochloropropane like many halocarbons can be measured in blood by gas chromatographic methods.
Treatment
Morgan (1989) gives 11 treatment steps on pp. 138-141.
10. PHOSPHINE (ALUMINUM PHOSPHIDE)
Toxicology
Phosphine gas is only slightly irritating to the respiratory tract, but is at least as toxic systemically as hydrogen cyanide. It is slowly released into treated produce or storage spaces by hydrolysis of solid aluminum phosphide (phostoxin). Mechanisms of toxicity are not well understood. Pulmonary edema is a common cause of death.
Chemical Effects
Phosphine is a colorless gas. Aluminum phosphide is usually formulated as a white pellet. The odor is said to resemble that of carbide or decaying fish. The odor threshold of the gas is about 2 ppm, but because of odorous impurities produced from metal phosphides,, the odor threshold for the pesticide is about 0.02 ppm.
Phosphine is spontaneously flammable if a trace of P2H4 is present. It combines violently with oxygen and halogens.
Exposure
Respiratory, oral
Symptoms/Signs
Morgan (1989) lists the principal manifestations of poisoning as fatigue, nausea, headache, dizziness, thirst, cough, shortness of breath, paresthesia, and jaundice.
Additional poisoning symptoms listed by Hayes (1991) include: lassitude, immobility followed by deepened restlesssness, ataxia, pallor, epileptiform convulsions, apnea, and cardiac arrest.
Laboratory
There are no practical tests for absorbed alkyl oxides, aldehydes, or phosphine that would be helpful in diagnosis of poisoning.
Treatment
Morgan (1989, pp. 138-141) gives general guidelines for treatment of
phosphine poisoning. He includes special emphasis: "Control CONVULSIONS.
Seizures are likely to occur in poisonings by phosphine."
Toxicology
Sulfuryl fluoride (Vikane® Gas Fumigant) is toxic to most living creatures including humans. It is colorless and odorless. Therefore it has no warning properties. It has an acute oral LD 50 in rats of 100 mg/kg. Symptoms and/or death in humans as a result of exposure to sulfuryl fluoride will depend on the concentration and the length of exposure experienced.
The mortality curve for acute inhalation exposure is very steep. Only a small margin exists between lethal and non-lethal exposures. The time/concentration relationship holds true: the higher the concentration, the faster the effect. It was not teratogenic in animal studies. Some signs of maternal toxicity (decrease body weight gain, increase water consumption) have been observed. Decreased fetal body weight indicates fetotoxicity.
Chronic, long term daily exposure may produce some tissue damage even at relatively low concentrations (>30 ppm). Fluorosis of the teeth may occur when humans are chronically exposed. Workers who frequently come into contact with sulfuryl fluorid gas can have their urine checked for fluoride.
Mutagenicity, carcinogenicity are currently under investigation. Mutagenic and carcinogenic effects of sulfuryl fluoride are presently unknown.
Chemical Effects
Sulfuryl fluoride is colorless, odorless, highly toxic gas with a very low vapor pressure (BP -67 F). It is packaged as a liquified compressed gas. At low concentration it is not irritating to mucous membranes. Therefore, it gives no warning of its presence. It has the ability to penetrate most fibrous or porous materials. This makes it an excellent broad spectrum fumigant because of its high toxicity and penetrating qualities. Skin and eye exposures to the liquid usually produce injury from freezing.
Exposure
Respiratory, eyes, skin. Human ingestion is highly unlikely since sulfuryl fluoride liquid turns to a gas at -67 F.
Symptoms/Signs
Persons exposed to sulfuryl fluoride may show little evidence of intoxication at first. Initial effects will probably be on the central nervous system. Central nervous system depression with slow speech and gait will be the first signs noted.
Symptoms of exposure to high concentrations: cause respiratory irritation, pulmonary edema, nausea, abdominal pain, CNS (central nervous system) depression, slowing of movements and speech, and numbness in the extremities. Exposures to progressively higher concentrations produce convulsions, tremors and strychnine-like muscular rigidity.
Laboratory
Urine Analysis for Fluorides: Urine samples from persons with potential overexposure to sulfuryl fluoride may be analyzed for total fluorides. However, the interpretation of these levels requires a base line (pre-exposure level) to determine if the increased fluorides is actually due to exposure to sulfuryl fluoride.
Treatment
Morgan (1989) does not make any special recommendations concerning sulfuryl fluoride. Follow the general guidelines for treatment of fumigants given in Morgan (1989) pp. 138-141.
The manufacturer of sulfuryl fluoride (Vikane® Gas Fumigant) gives more information on treatment in bulletins and labels. They also provide an emergency telephone number to call in case of poisoning.
Emergency Telephone Number: (517) 636-4400.
There is no known antidote. Clinical observation is essential. Treatment is based on the clinical judgement of the physician and the individual reaction of the patient. For emergency or further information, call the emergency number (517) 636-4400.
For Emergencies or Further Information Call (517) 636-4400
The manufacturer provides the following general information for physicians:
"If the patient is removed to fresh air and put at rest, central nervous
system symptoms and signs will be the first to appear. it is essential
to keep such an individual at bed rest for at least 24 hours. Clinical
observation should be directed at the pulmonary, hepatic, and renal systems.
A post mortem finding in one fatality attributed to Vikane was pulmonary
edema, with death attributed to cardiorespiratory failure. Convulsions
may ensue with respiratory arrest being a terminal event. Assisted respiration
may be necessary."
FUMIGANTS
| CHEMICAL TYPE | BRAND | CHEMICAL | SITE-PEST | Morgan (1989) |
| Hydrocarbon | MOTH BALLS
MOTH FLAKES |
NAPHTHALENE | CLOTHES MOTHS | pp. 138-142 |
| Chlorinated hydrocarbon | BORER CONTROL
MOTH CRYSTALS MOTH TEK DEODORIZER ROSE PARA MOTH |
PARADICHLOROBENZENE | LILAC BORER
PEACH BORER LESSER P. BORER CLOTHES MOTHS APPLE BORER |
pp. 134-141 |
| Chlorinated hydrocarbon | VERTIFUME
GRAIN FUMIGANT PREMIUM GR. FUMIGANT |
CARBON TETRACHLORIDE | STORED PRODUCT PESTS | pp. 138-142 |
| Carbon disulfide | GRAIN FUMIGANT
PREMIUM GR. FUMIGANT |
CARBON DISULFIDE | STORED PRODUCT PESTS | pp. 138-143 |
| Cyanide | M-44 COYOTE GETTER
CYANOGAS CYANAMID FUMIGANT CYCLON |
HYDROGEN CYANIDE | STORED PRODUCT PESTS
COYOTES |
pp. 143-145 |
| Chloropicrin | LARVACIDE
PICFUME TELONE C-17 |
CHLOROPICRIN | STORED PRODUCT PESTS
SOIL PESTS |
pp. 138-141 |
| Chlorinated hydrocarbon | TELONEII
TELONE C-17 D-D VIDDEN-D |
DICHLOROPROPENE &
DICHLOROPROPANE |
SOIL PESTS | pp. 138-141 |
| Halogenated hydrocarbon | BROM-O-GAS
BROM-O-SOL DOWFUME MC-2 DOWFUME MC-33 TERR-O-GAS |
METHYL BROMIDE | STORED PRODUCT PESTS
DRYWOOD TERMITE SOIL PESTS WOOD BORING BEETLES |
pp. 142 |
| Inorganic | VIKANE | SULFURYL FLUORIDE | DRYWOOD TERMITE
STORED PRODUCTS WOOD BORING BEETLES |
pp. 138-141 |
| Phosphorus compound | PHOSTOXIN
GASTOXIN FUMITOXIN |
ALUMINUM PHOSPHIDE
(PHOSPHINE GAS) |
STORED PRODUCT PESTS | pp. 138-141 |
| Inorganic | GRAIN FUMIGANT
PREMIUM GR. FUMIGANT |
SULFUR DIOXIDE | STORED PRODUCT PESTS | pp. 138-141 |
| Halogenated hydrocarbon | DBCP
FUMAZONE NEMAGON NEMAFUME NEMASET |
DIBROMOCHLOROPROPANE | SOIL PESTS | pp. 138-141 |
| Aldehyde | ACRITET
CARBACRYL |
ACROLEIN
(ACRYLALDEHYDE) |
SPACE FUMIGANT | pp. 138-141 |
| Halogenated hydrocarbon | BROMOFUME
EDB NEPHIS |
ETHYLENE DIBROMIDE | SOIL PESTS | pp. 138-141 |
| Oxide | ETO
OXYRANE |
ETHYLENE OXIDE | SPACE FUMIGANT | pp. 138-141 |
| Thiocarbamate | VAPAM
NEMASOL |
METAM SODIUM | SOIL PESTS | pp. 98-99 |
| Thiadiazone | BASAMID
DAZOMET |
DAZOMET | SOIL FUMIGANT | (800)832-4357
(800)832-HELP |
Table of Contents | Preface
| Section I | Section II
| Section III | References